Abstract
Cell therapy manufacturing represents one of the most complex production environments in modern biotechnology. Predictive analytics, intelligent automation, and quality governance capabilities can improve manufacturing resilience, compliance, and scalability when implemented within validated, regulated frameworks. This publication examines the role of data systems across cell therapy production ecosystems, with emphasis on process optimization, data architecture, quality systems, risk management, and responsible validation within regulated environments.
1. Introduction
Cell therapy manufacturing sits at the intersection of biotechnology, precision medicine, and advanced manufacturing engineering. Unlike conventional pharmaceuticals that rely on standardized chemical inputs and repeatable batch synthesis, many cell therapies operate with patient-specific starting material and high-dimensional biological variability. Minor fluctuations in handling time, temperature exposure, or process timing may propagate into meaningful differences in cell yield, viability, phenotype, and potency. These dynamics place manufacturing and quality systems under continuous pressure.
Data systems are relevant because they can model complex relationships between process signals and outcomes, enabling earlier detection of risk and more consistent decision support. Analytical tools do not replace accountable human oversight in Good Manufacturing Practice environments. Instead, they augment manufacturing scientists, engineers, and quality professionals by expanding analytical coverage, accelerating review, and improving consistency of routine judgments when supported by validated controls.
2. Scientific Foundations of Cell Therapy
Cell therapy uses living cells as the active therapeutic agent. Therapeutic cells can proliferate, differentiate, and respond adaptively to biological cues. This differentiates them from small molecule drugs and many protein biologics, which function through comparatively static chemical interactions.
Modern cell therapies include unmodified cellular products as well as engineered systems in which cells are activated, expanded, or genetically modified outside the body and then returned to the patient. The living nature of the product introduces quality attributes that are distinct from traditional pharmaceuticals, including viability, phenotype stability, and functional potency. These attributes are tightly coupled to manufacturing conditions and therefore demand robust process control and data integrity.
3. Therapeutic Mechanisms and Clinical Applications
Cell therapies achieve clinical effect through dynamic biological interaction. In oncology, engineered immune cells can recognize malignant targets and initiate targeted cytotoxic activity, often with the ability to persist and expand after administration. In regenerative medicine, stem cell-based approaches aim to repair damaged tissues through differentiation and paracrine signaling. In autoimmune disease, immunomodulatory cell populations are investigated for their ability to restore immune balance.
These mechanisms imply that manufacturing is inseparable from clinical function. Process choices that influence activation strength, expansion kinetics, or cellular phenotype can alter therapeutic performance. Therefore, any optimization strategy must be grounded in clinically meaningful quality attributes and scientifically justified control strategies.
Find Active Clinical Trials
Federally registered cell therapy and oncology trials are searchable through ClinicalTrials.gov, covering CAR-T, stem cell, and immunotherapy research with open enrollment status.
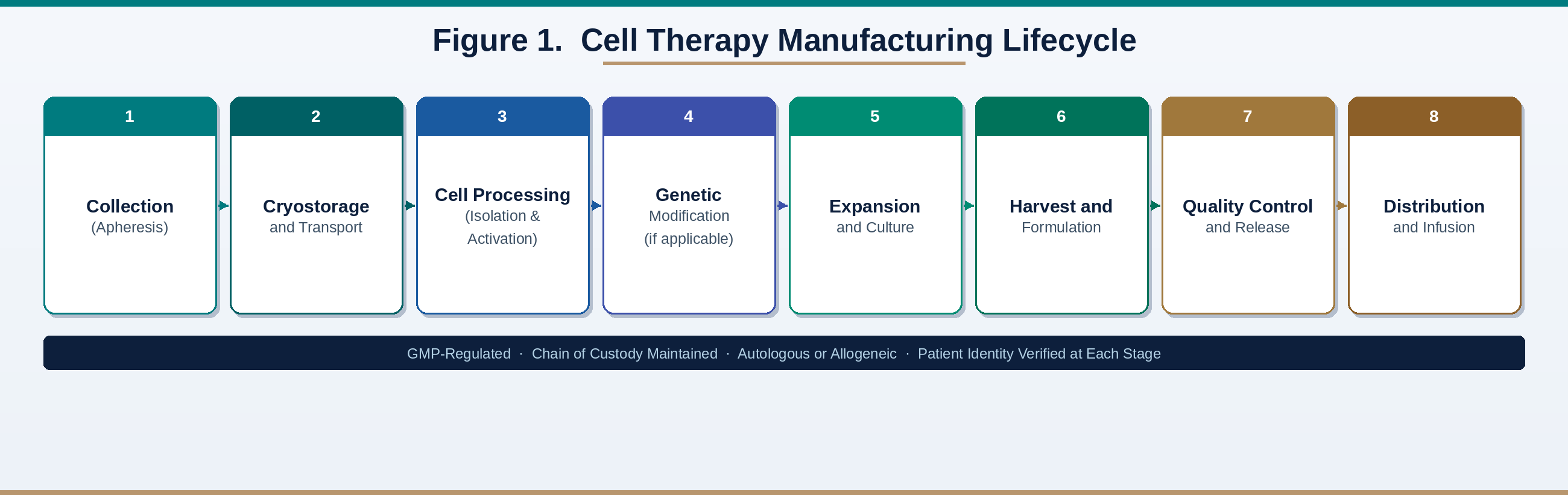
4. Manufacturing Complexity in Cell Therapy Production
Cell therapy manufacturing typically begins with material acquisition, followed by controlled transport, cell processing, optional genetic modification, expansion culture, harvest and formulation, quality control testing, and distribution to the clinical site. For individualized products, each batch corresponds to a single patient, which creates parallel production streams rather than high-volume pooled batches.
This operating model introduces scheduling complexity, heightened contamination sensitivity, extensive documentation, and high-consequence deviation management. Because the product is living, it cannot be terminally sterilized in the same way as many pharmaceuticals. A strong quality system therefore relies on process controls, environmental monitoring, operator training, and rapid detection of abnormal signals. Data systems become valuable when they can convert high-volume operational data into actionable signals for risk prediction, review prioritization, and process learning.
5. Autologous and Allogeneic Manufacturing Systems
Autologous manufacturing uses cells sourced from the same patient who will receive the therapy. The defining advantage is immunological compatibility, since the product originates from the patient. The defining challenge is variability. Starting material differs across patients in cell composition, viability, activation response, and expansion potential. Manufacturing capacity must be scheduled for individualized lots, and chain-of-identity controls must be exceptionally strong.
Allogeneic manufacturing uses cells from a donor source to create product intended for multiple recipients. This model supports larger-scale batch production and, in principle, more standardized starting material. However, it introduces its own scientific and operational challenges, including immune compatibility management, batch release strategy across recipients, and supply planning for inventory.
Analytical opportunities differ by model. In autologous systems, predictive analytics can focus on early indicators of batch risk, patient material quality, and cycle time forecasting. In allogeneic systems, data tools can support scale optimization, inventory planning, and enhanced process control at higher throughput. In both cases, model validation must align with regulated change control, data integrity expectations, and the need for explainable decision support.
6. Data Architecture for Manufacturing Readiness
Manufacturing systems are only as reliable as the data architecture that supports them. Cell therapy production generates heterogeneous data types that include batch records, equipment telemetry, environmental monitoring results, QC assay outputs, and operator logs. Effective analytical infrastructure requires harmonizing these data types into a coherent, queryable representation that preserves context, timestamp accuracy, and chain-of-custody integrity.
Effective implementation often follows a layered approach. The operational layer captures raw signals. The integration layer harmonizes data across MES, LIMS, and QMS platforms using common identifiers and time alignment. The analytics layer supports feature engineering and model development. The governance layer ensures that decisions remain traceable, reviewable, and compliant. This architecture enables analytical systems to provide value without compromising accountability.

7. Quality Systems and Compliance in Regulated Manufacturing
Quality systems apply analytics to strengthen detection, investigation, and documentation of quality events within regulated manufacturing. In cell therapy manufacturing, quality operations must address high-consequence risks such as contamination, identity errors, equipment excursions, and process deviations. These risks are managed through a combination of procedural controls, environmental monitoring, training, documentation, and formal quality system processes including deviation management and corrective and preventive actions (CAPA).
Analytical tools can support quality activities when implemented as decision support within a validated governance framework. One useful pattern is signal triage — manufacturing produces large volumes of time-stamped data streams that include incubator conditions, temperature histories, particle counts, microbial monitoring results, and equipment alarms. Detection models can learn normal operating ranges and flag abnormal combinations of signals earlier than manual review, enabling faster containment and more timely investigation.
A second pattern is review prioritization for electronic batch records and quality event documentation. Quality professionals often face large documentation workloads that include routine checks, exception reviews, and cross-referencing of supporting evidence. Structured classification models can group events by similarity, suggest likely root cause categories, and highlight missing fields or inconsistent timestamps — reducing friction while preserving the quality unit's professional judgment.
Despite these benefits, analytical tools applied to quality must satisfy strict governance requirements. Regulated manufacturing emphasizes data integrity, auditability, and change control. Models used in quality contexts should have clearly defined intended use, documented training datasets, performance metrics tied to the use case, and monitoring plans that detect drift. A stable governance model also requires version control for training data, code, and model parameters, along with procedures for revalidation when the model is updated.
A practical implementation pathway begins with low-risk augmentation. Organizations can first deploy analytical tools for internal trend detection and workload prioritization, then expand to more integrated use once data pipelines, validation practices, and operator training mature. This staged approach aligns adoption with the principles of a pharmaceutical quality system, ensuring that new capabilities strengthen control rather than eroding it.
External Resources
The following authoritative sources informed this publication and are provided for further reading.
FDA — AI in Drug Manufacturing
Discussion paper on data systems in regulated manufacturing environments.
FDA.gov ↗ClinicalTrials.gov — Cancer
Search active and completed cancer and cell therapy trials by condition.
ClinicalTrials.gov ↗NCI — Clinical Trials
National Cancer Institute guide to participating in cancer research studies.
Cancer.gov ↗ASCO — Clinical Oncology
American Society of Clinical Oncology — oncology research and guidelines.
ASCO.org ↗PubMed — Cell Therapy Research
Search peer-reviewed literature on cell therapy and bioprocess science.
PubMed ↗ICH Q10 — Quality System
International Council for Harmonisation pharmaceutical quality system guideline.
ICH.org ↗Download the full publication
Complete PDF with all figures and references.